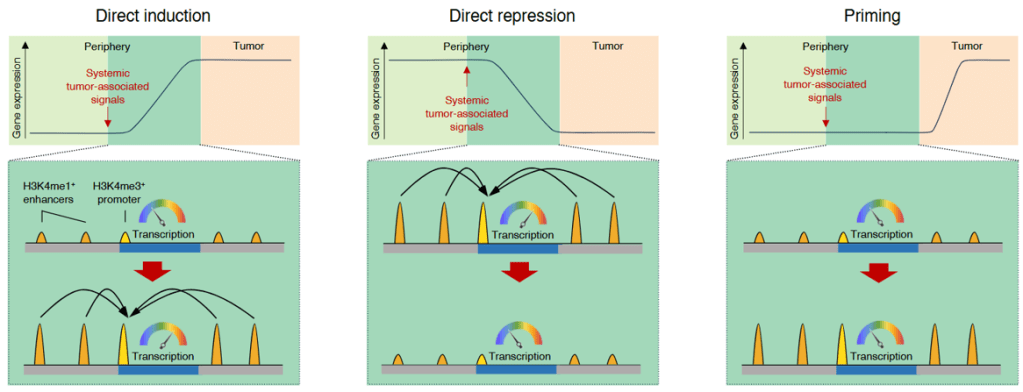
Abstract. Monocytes are recruited to tumors and undergo transcriptional reprogramming resulting in tumor-promoting functions. It remains unknown whether systemic tumor-derived signals could alter the epigenomic landscape of peripheral monocytes before they reach the tumor, thus shaping their subsequent response to the tumor microenvironment. Here, we performed genome-wide analyses of multiple histone modifications (H3K4me1, H3K4me3, H3K27ac) and chromatin accessibility in a mouse model and found that the distal tumor caused extensive remodeling of both H3K4me3+ promoters and H3K4me1+ enhancers in peripheral monocytes. Specifically, this involved the repression of interferon-responsive promoters and enhancers as well as the establishment of enhancers harboring binding motifs for transcription factors downstream of inflammatory signaling pathways. Overall, these results indicate that the epigenomic landscape of peripheral monocytes is altered in response to a distal tumor, and this can shape the transcriptional response of monocytes to the tumor microenvironment.
Authors. Máté Kiss; Laszlo Halasz; Eva Hadadi; Wilhelm K Berger; Petros Tzerpos; Szilard Poliska; Daliya Kancheva; Aurélie Gabriel; Romina Mora Barthelmess; Ayla Debraekeleer; Jan Brughmans; Yvon Elkrim; Liesbet Martens; Yvan Saeys; Bence Daniel; Zsolt Czimmerer; Damya Laoui; Laszlo Nagy; Jo A Van Ginderachter.